TESTING BATTERY SYSTEMS
Storage batteries are becoming increasingly important in terms of power supply security. However, they’ve been in use for many decades, especially – although not only – in the field of emergency power supply. But batteries are subject to irreversible, unavoidable aging processes during the course of time, which result in reduced available capacity. In order to ensure the availability of a stationary battery system’s rated capacity, periodic testing and well-organized maintenance are necessary. The goal is to ascertain the battery’s current condition and to pinpoint aged blocks before major damage occurs, thus impairing battery capacity.
THE BATTERY
The term "battery" is derived from military parlance where it designates an assembly of several guns. Analogously, the term was adopted to describe an aggregate of several electrically connected galvanic cells. As of the second half of the 20th century, use of the term "battery" was also extended to include individual primary or secondary cells, although the term "accumulator cell" or "rechargeable battery" is more commonly used for the latter. The aforementioned shift in linguistic usage was addressed in DIN standard 40729, Accumulators; galvanic secondary cells; general terms and definitions, which defines a battery as "several connected cells", but this differentiation in terminology has become blurred in everyday usage.1
"Battery" is the generic term for energy storage devices, as well as the designation for any primary battery. Non-rechargeable batteries are called primary batteries. Batteries that can be recharged are called secondary batteries or rechargeable batteries in everyday language, and are also known as accumulators.2
Starter batteries for motor vehicles, traction batteries or deep cycle storage batteries for electric vehicles and stationary applications such as uninterruptible power supplies are always accumulators.
Appliance batteries are used to power small, mostly portable devices like watches, radios, toys, flashlights and the like, as well as permanently installed appliances such as smoke detectors. Standard designs are used in most cases.
Device batteries must be compact, suitable for use in any orientation, lightweight and yet mechanically robust. They mustn’t leak or outgas during normal storage and use in the respective device. They’re commercially available in numerous variants based on zinc-carbon or alkaline-manganese materials. Availability of zinc-carbon batteries has been decreasing since the 2000s, and they’re hardly manufactured at all anymore.
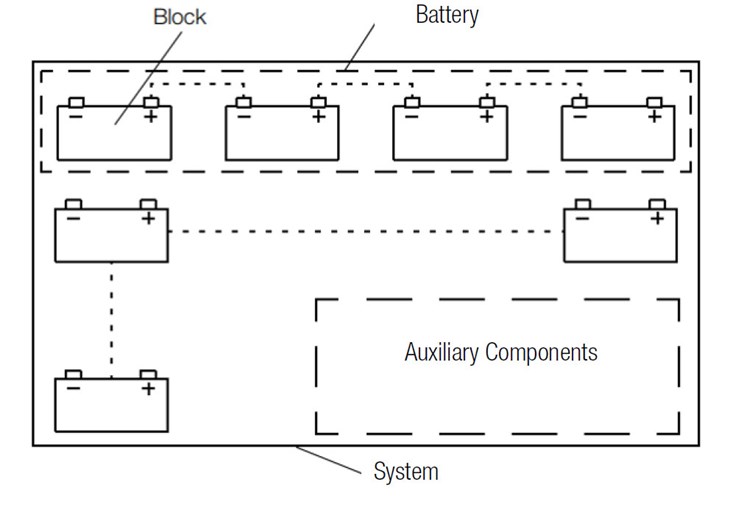
Stationary Battery Layout
RELEVANT MEASUREMENTS
- Alternating and direct voltage
- Direct current
- Internal Resistance
- Acid Density
- Temperature
FLOAT VOLTAGE:
All voltage values of the individual battery blocks within a battery system are recorded, often at mid-range intervals (inspection every 3 months).
CHARGING/DISCHARGING:
Block voltage values are recorded several times to this end, for example during a controlled discharge process. At least two test runs must be completed.
RESISTANCE:
In addition to measured values for block voltage (see FLOAT VOLTAGE), the associated internal resistance values of the battery blocks are also logged.
INTERVAL U / INTERVAL U + I:
Voltage and current curves during charging and discharging can be recorded at freely definable time intervals.
CONNECTORS:
Measurement of voltage drop across the connectors – also during discharge
TEMPERATURE:
Acquisition of block temperatures – frequently carried out together with float voltage measurement
DENSITY:
Density measurement of the electrolyte in the battery blocks (for vented batteries)
1 Dirk Flottmann, Detlev Forst, Helmut Roßwag: Chemie für Ingenieure: Grundlagen und Praxisbeispiele
2 zvei.org, October 2019